Abstract
Introduction:
Axicabtagene ciloleucel (axi-cel) and tisagenlecleucel (tisa-cel) are CD19-directed autologous chimeric antigen receptor (CAR) T-cell (CART) therapies approved for the treatment of adults with relapsed or refractory (R/R) large B-cell lymphoma (LBCL). Less than 25% of patients on the initial registration trials were over the age of 65. Real world data on the survival and toxicity outcomes of older patients receiving CAR T-cell (CART) therapy are limited. We used the CIBMTR registry to study the impact of age on response, survival outcomes and incidence of toxicity after receiving commercial CART.
Methods:
CIBMTR registry data were analyzed for clinical data on adults ≥ 18 years with a diagnosis of LBCL who received commercial CART from May 2018 to June 2020. Cumulative incidence and severity of cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS) were reported as per the consensus ASTCT criteria and further described with univariate logistic regression analyses. Efficacy and safety outcomes were assessed using age as a continuous variable and amongst four age groups: 18-54, 55-64, 65-74, and ≥75 years. Multivariable analysis was performed using Cox proportional hazards models using hazard ratio (HR) and 95% confidence intervals (CI) and was adjusted for gender, performance score, HCT CI, time from diagnosis to CART infusion, disease status at infusion, prior lines of therapy, bridging therapy, and CAR T cell product.
Results:
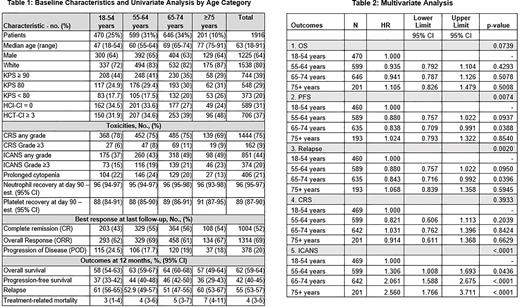
Across 97 centers, 1916 patients with LBCL received axi-cel (1438 (75%)) or tisa-cel (481 (25%)). Patient characteristics are summarized in table 1. Overall, the median age at infusion was 63 years (18 - 91 years), with 44% of patients over the age of 65, of whom 51% had a KPS <90 and 41% had HCT-CI >3. Diffuse LBCL not otherwise specified, germinal center B-cell subtype, activated B-cell subtype and double/triple-hit represented 25%, 34%, 24%, and 14%, respectively, with 2.7% of patients having CNS involvement. About 40% of patients had primary refractory disease and 76% of patients had ≥ 3 prior lines of treatment. Twenty six percent of patients had a prior autologous stem cell transplant (auto-SCT), 27% were 65-74 years and 10% were ≥ 75 years. Twenty three percent of patients received bridging therapy, 78% with systemic therapy and 27% with radiotherapy.
The rate of any grade of CRS was 75%, ranging from 78% to 69% with all age groups, and ICANS was 44%, ranging from 37% to 49%, respectively. The 12-month OS, PFS, and relapse rate was 61.6% (59.4-63.9), 42.2% (39.9-44.5), 54.9% (52.5-57.2), respectively (Table 1).
In multivariate analyses (MVA) analyzing the impact of age on outcomes, age as a continuous variable did not impact OS (p=0.54), PFS (p=0.07), relapse (p=0.09) and any grade of CRS (p=0.87). A non-linear association between an increase in age with PFS and relapse was observed, with better PFS and decreased relapse in patients in the 65-74 years age group (PFS: HR 0.84, 95% CI (0.71-0.99), p=0.04, relapse: HR 0.84, 95% CI (0.72-0.99), p=0.04) compared to patients aged 18 to 54 years (Table 2). An association of increased risk for any grade of ICANS with annual increase in age (HR 1.03, p<0.001) was identified. Compared to patients aged 18 to 54 years, a linear association between age and any grade of ICANS was also demonstrated in age categories (55-64 years (OR 1.31, 95% CI (1.01-1.69), p=0.04), 65-74 years (OR 2.06, 95% CI (1.59-2.68), p<0.0001), and ≥75 years (OR 2.56, 95% CI (1.77-3.71), p<0.0001)).
Conclusions:
This retrospective CIBMTR registry data analysis demonstrates that CD-19 CAR T-cell therapy is feasible for older adults and that advanced age does not impact mortality. Increasing age was significantly associated with the incidence and grade of ICANS, but no relationship with age was found for CRS. One age group demonstrated better PFS and less relapse compared to younger patients with statistical significance but this may represent a degree of bias in selection of older patients. Advanced age should prompt anticipatory management of neurotoxicity and comorbidities may impact outcomes after CAR T-cell therapy.
Disclosures
Ahmed:Chimagen: Consultancy, Research Funding; Servier: Membership on an entity's Board of Directors or advisory committees; Myeloid Therapeutics: Consultancy; Xencor: Research Funding; Merck: Research Funding; Seagen: Research Funding; Tessa Therapeutics: Consultancy, Research Funding. Turtle:GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees; Kite Pharma, a Gilead Company: Membership on an entity's Board of Directors or advisory committees; Expert Connect: Consultancy; Decheng Capital: Consultancy, Membership on an entity's Board of Directors or advisory committees; Century Therapeutics: Membership on an entity's Board of Directors or advisory committees; Precision Bioscience: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Arsenal Bio: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Myeloid Therapeutics: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Eureka Therapeutics: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; T-CURX: Membership on an entity's Board of Directors or advisory committees; Caribou Bioscience: Current holder of stock options in a privately-held company, Membership on an entity's Board of Directors or advisory committees; Nektar Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Juno Therapeutics, a BMS Company: Patents & Royalties, Research Funding; Allogene: Membership on an entity's Board of Directors or advisory committees; Prescient Therapeutics: Membership on an entity's Board of Directors or advisory committees. Pasquini:Novartis: Research Funding; Bristol Myers Squibb: Consultancy, Research Funding; Kite: Research Funding; Janssen: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal